Batch variability of mRNA jabs, quality control, and the placebo theory
Scientists from Denmark and Germany are making some startling discoveries that leave us with more questions than answers
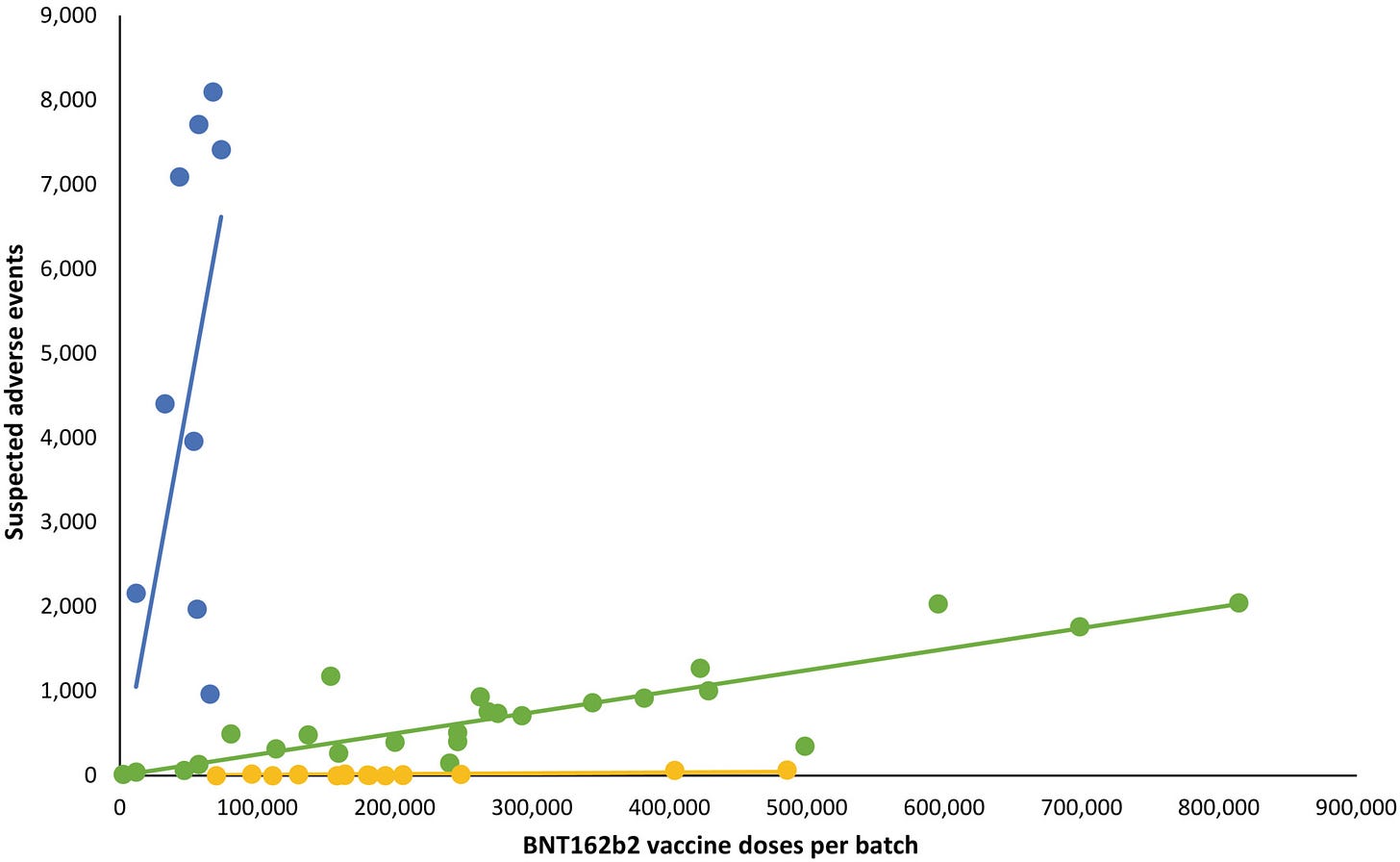
About a month ago, some Danish scientists published a study in European Journal of Clinical Investigation showing how certain batches of Pfizer’s mRNA injections resulted in significant serious side effects, while the adverse reaction profile of other batches resembled that of a placebo. The chart below shows the risk profile of the various batches and how many doses of each batch were given in Denmark. The most risky batches are marked in blue, those less risky in green, and the most harmless in yellow.
(For reference, a full batch size is approximately 1.4 million doses)
Some German scientists investigated further and found that while the blue and green batches were monitored by the Paul Ehrlich Institute, Germany’s medical regulatory body, the yellow ones were not. Since BioNTech is a German company, PEI is the regulatory body that would have ensured the safety of the medical products before they were shipped to the rest of Europe.
Prof. Dr. Gerald Dyker and Prof. Dr. Jörg Matysik discussed their concerns about the mRNA jabs in a fascinating interview in Punkt.PRERADOVIC.
Dr. Dyker presents three possibilities for the discrepancies between the batches.
BioNTech and PEI released the initial batches, found out how dangerous they were, and quietly halted their distribution. That would explain why the sizes of the blue batches are much smaller than those of the green and yellow.
The manufacturers released the first batch as an experimental part of their process optimization.
The first batches were indeed distributed throughout Europe, and Denmark only received a relatively small portion of the dangerous batches.
Dr. Dyker also points out that the safe batches marked in yellow resemble those of a placebo. Keep in mind that in the EU, a placebo is not necessarily a saline solution. It only needs to lack the active ingredient in order to qualify as a placebo. Dyker hypothesizes that since PEI was not monitoring these batches, they may have already known they were placebos, and thus assumed no need to check them. Of course there is no way to know this for sure based on the data, which is why the German scientists have sent inquiries to PEI, to no avail.
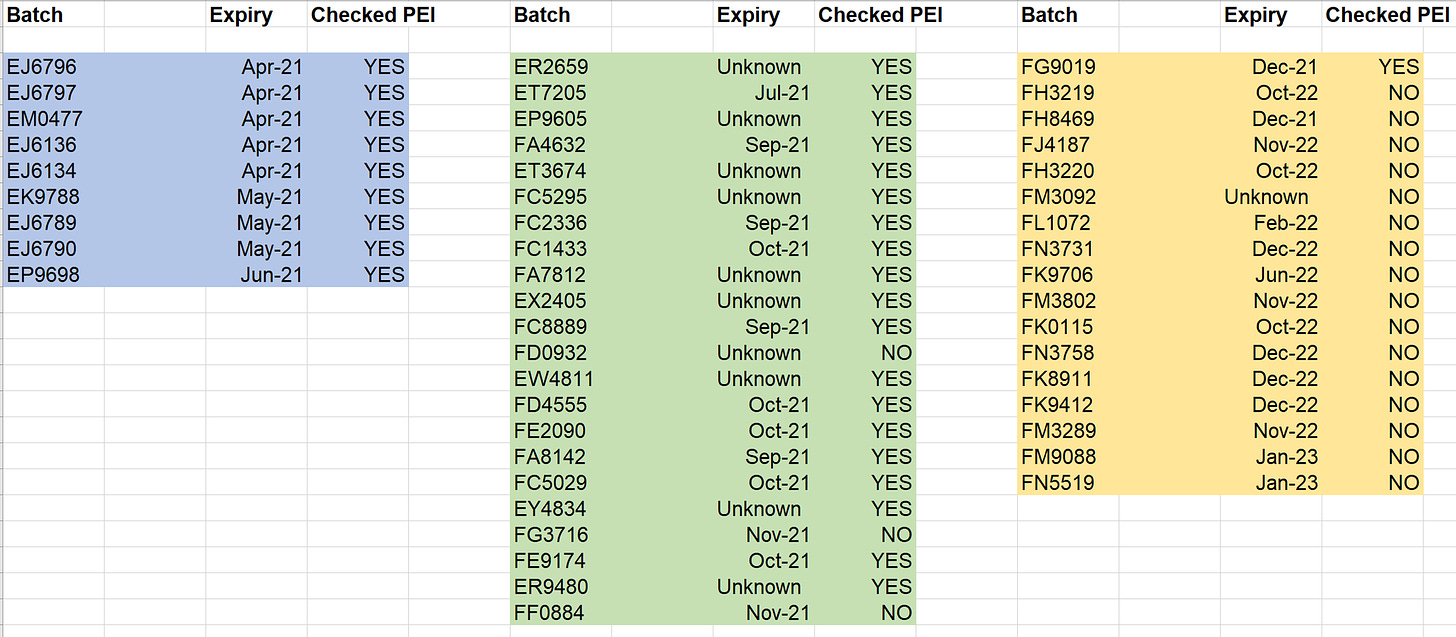
Missing from the discussion were the expiration dates of the various batches. The professors seem to imply this in their discussion, but they fail to explicitly point out that the dangerous batches marked in blue were given in the spring of 2021. The green batches were delivered in fall, and the yellow primarily in fall and winter.
Critics have rightly pointed out that the study does not take confounding factors into account, such as the time of delivery or the age profile of the recipients. Since the elderly and frail were the ones primarily receiving the early batches, and since their risk of death and disease is already higher, that could skew the frequency of adverse events in these batches. Healthy people likely received the yellow batches, and they were more likely to resist any potentially adverse effects from the jabs. Critics also point out that the smaller proportion of the blue batches in Denmark skews the numbers to appear more dangerous than they actually might be.
While the criticism regarding confounders is valid, it is not unrealistic for the first batches to be of far lower quality and more dangerous than subsequent batches. Anyone who works in the developing and manufacturing of advanced technological products can tell you how difficult it is to ramp up production of a new product without running into glitches. Even with an enormous amount of safety testing, process optimization, and quality control, it takes time to iron out mistakes. And if there are any mistakes in the manufacturing, logistical, and delivery processes, they are most likely to be apparent shortly after the first shipment.
This is the case for any new product. And I don’t care how amazing your company is or how brilliant you are. Ramping up production from 0 to billions within a year WILL result in a quality control failure. It is absolutely going to happen.
So while it may be that the high rate of adverse events shortly after the rollout is due to the frailty of the patients receiving the jab, it seems much more likely to me that it was the result of a quality control failure. In their interview, Drs. Dyker and Matysik point out some of these potential points of quality failure in the mRNA products.
Some have taken the placebo claim as evidence of the pharmaceutical companies running an experiment on the public.
That seems to be a bit of a stretch, and the expiration dates of the batches do not support this theory. More likely, the first batches were dangerous, and the manufacturers quickly fixed any obvious problems in the production process. If they did in fact use a placebo, it would not have been to run an experiment, but to try and deliver a product later in the year that they knew was not going to cause so much damage.
In the end, this is all speculation. The Danish study has its flaws, but no matter how you slice it, the mRNA batches resulting in adverse events do not look good. Maybe the various theories about the data are wrong, but the burden of proof is not on researchers to explain what is going on. That lies with the manufacturers and the regulators. And there is no way of knowing for sure the intentions of the manufacturers and regulatory agencies until BioNTech and PEI provide the answers.
Until that time comes, we have to keep questioning.